|
The intermolecular forces for the ether are the relatively weak dipole-dipole interactions that do not draw the molecules together as tightly as hydrogen bonds would. Other liquids, such as diethyl ether, do not demonstrate strong surface tension interactions. describes the internal friction of a moving fluid. hardness, elasticity, tensile strength, ductility, brittleness, viscosity, density, malleability, buoyancy, pressure. 
What is achieved by the bodys regulation of sodium ions, proteins, and red blood cells Study with Quizlet and memorize flashcards containing terms like Viscosity, Osmolarity, Edema and more. What occurs when osmolarity is low, therefore blood pressure drops Optimum Osmolarity. if the solid mixes to form a homogenous substance with the liquid then the solid is soluble. Viscosity is a measure of a fluids resistance to flow. total particles that CANNOT pass through a blood vessel wall. (B) Surface tension allows a paper clip to float on water's surface. A solid crystal or a solid powder which absorbs water from the atmosphere. 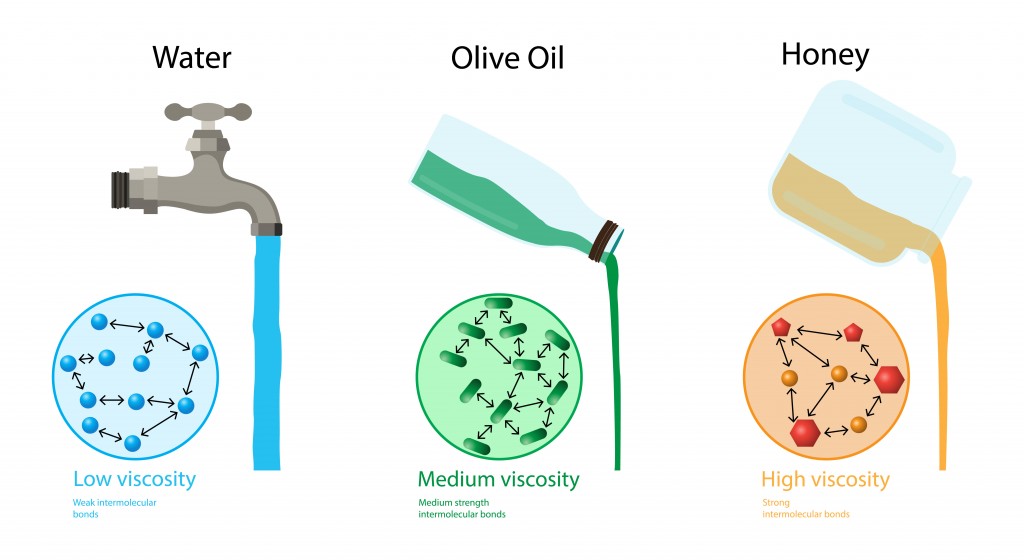
Poiseuille’s law for flow in a tube is Q (P2 P1)r4 8l. For laminar flow in a tube, Poiseuille’s law for resistance states that R 8l r4. 
Flow is proportional to pressure difference and inversely proportional to resistance: Q P2 P1 R. \): (A) Molecules at the surface of a liquid are pulled downwards into the liquid, creating a tightened surface. Viscosity has units of (N / m2) s or Pa s.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
